
If you’re thinking about starting a small business, you have to have passion. But don’t let passion eclipse your business acumen.
Your idea to start a specific business in a particular city is only as good as the research you’ve done. Supply and demand are driving factors for business success, and if your city is already overserved by your planned business type, then you could be on a dead-end path. It’s time to change something, whether it’s a shift in location or a change of business plans.
Americans opened 839,978 new businesses last year. And, despite the reports, they weren’t all Dollar Generals. Unfortunately, around 20% of new businesses will be gone within two years. After five years, barely half will remain.
ZenBusiness wants to tip the odds in your favor. We figured out the types of businesses most in demand in the 50 biggest cities in the U.S. These are the “best business types” to start: the trades that get the most Google searches relative to the number of local businesses already catering to that demand.
Key Findings
- Restaurants of different types are the best businesses to start in 17 cities.
- However, if different restaurant types are considered separate categories, the gym comes out on top with 15 cities, including NYC and LA.
- Averaged across the 50 biggest cities, the fitness industry has the most demand compared to supply (55.1 searches per business).
- In Las Vegas, there are 1,009 monthly searches for Indian food per local restaurant – the highest demand/supply disparity in our study.
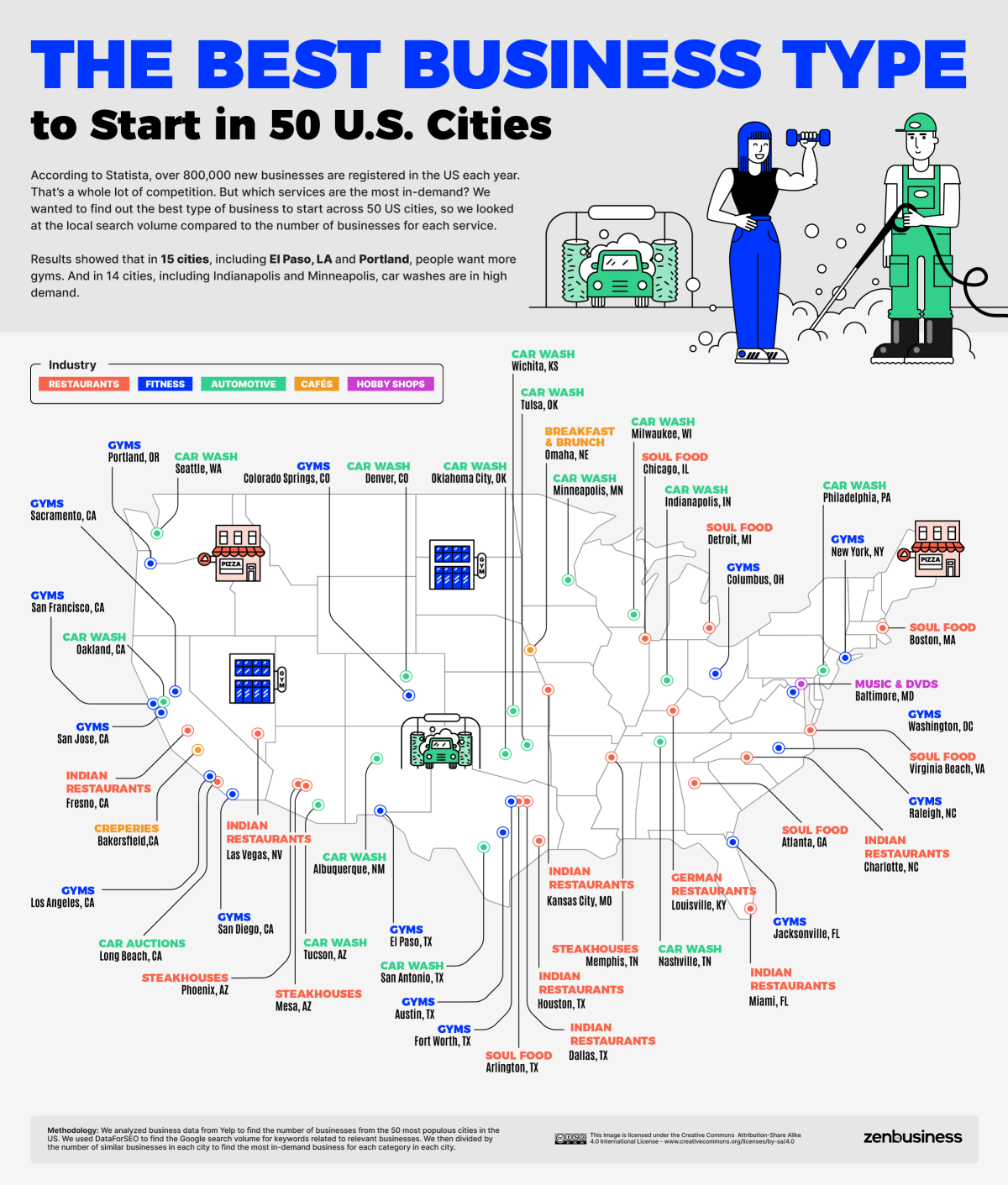
Demand for Gyms, Car Washes, and Eateries are Highest
American cities are hungry. Who’s going to feed them? Google searches for food are the highest compared to the number of relevant businesses in 17 big cities, including Chicago, Las Vegas, and Detroit. Seven of these cities are most in want of Indian food. In fact, people in Las Vegas search for Indian restaurants 1,009 times per local restaurant per month, the biggest supply/demand disparity in our study.
Click here to see this image in full size
But if you consider restaurant specialties as separate business types, the gym takes over as the most common best business – with searches most outstripping availability in 15 cities. Gyms are booming thanks to a culture shift towards self-care and lower-budget memberships.
However, you still need to know your locality: the demand for gyms compared to supply is twenty times greater in Fort Worth than in Long Beach, where a reasonable balance has already been reached.
From Pizza to Piercing: The Most In-Demand Industries Across American Cities
Next, we aggregated our results across the 50 most populous U.S. cities to get a broader look at how America is trying to shop. Pizza gets the most searches altogether (1,652,000), although it is only the fourth-best business to start since there are already 14,743 pizzerias across our sample area.
Pizza is known for its options, and the same goes for the pizzeria. If you’re hungry for a slice of the pizza trade, your first decision is whether to franchise an existing brand or go it alone. You also need to decide what combo of eat-in, collect, or delivery to go for and which extras and alternatives to offer. And then there’s the menu…
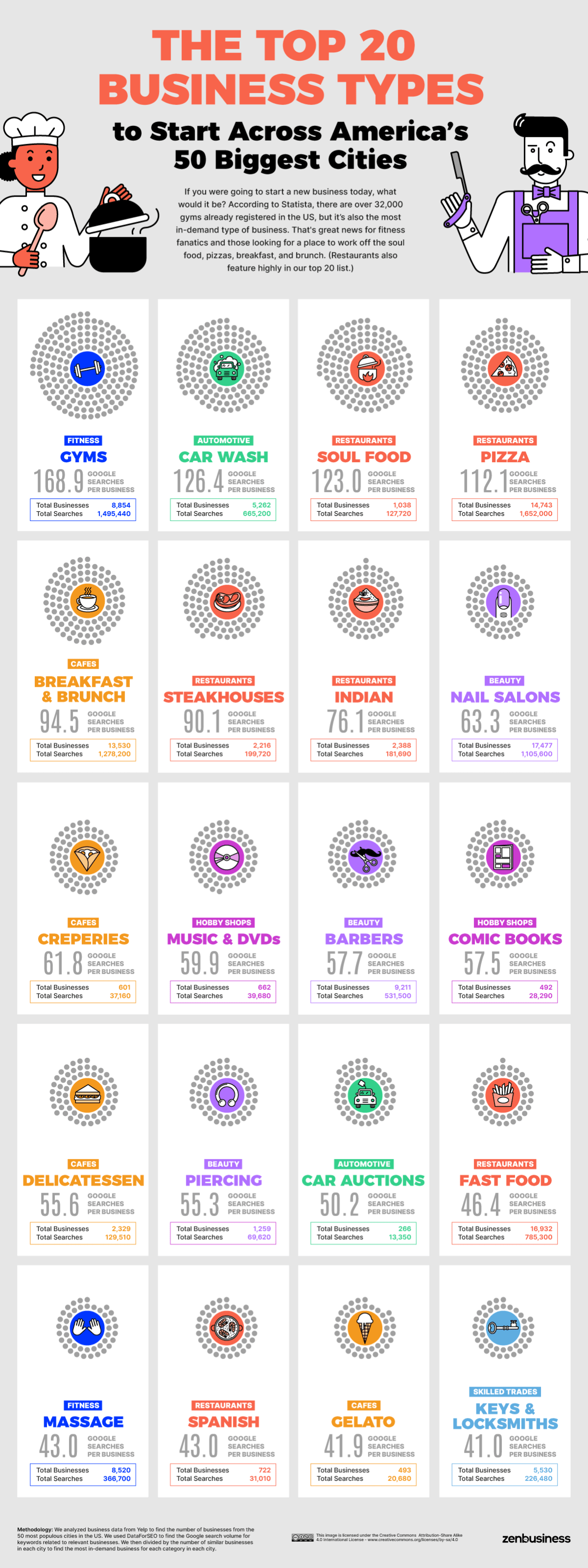
If you dream of becoming Comic Book Guy (or Gal), then the good news is that this business type is viable across much of urban America (at least more so than a Leftorium or Mr. Plow). Comic stores have a low search volume across all 50 cities in our study (28,290), but with just 492 businesses existing to answer these needs, there is a promising demand/supply rate. The world of comics is cast with strong (if morally dubious) entrepreneur role models to inspire you.
Supply and demand shouldn’t be your only concerns when starting a business. Pair our industry-specific tool with Inc’s broader guide to the cities where factors like job creation, wage growth, and funding deals are booming. But do make sure to study our stats and conduct your own research into local interest for your preferred product or service. As our research proves, in many U.S. cities, demand is greater than you’d think.
METHODOLOGY & SOURCES
We pulled popular business types from Yelp’s Categories and grouped them into industries. The number of businesses from each category within America’s 50 most populous cities (taken from the U.S. Census Bureau) was found using Yelp Fusion API. For each business type, keywords were formulated by adding terms such as “restaurants,” “stores,” “shops,” “parlors,” etc., depending on the type of business.
We then collated Google search volumes for each keyword related to a specific business in each city type using DataForSEO, using the term “near me,” e.g., “Indian restaurants near me.” Competition in each business type in each city was estimated by dividing the number of searches for that business by the number of businesses of that type in that city. The final dataset spans 159 business types across 14 industries and 50 most populous cities in the United States.
The data was gathered in February 2022.
Disclaimer: The content on this page is for information purposes only and does not constitute legal, tax, or accounting advice. If you have specific questions about any of these topics, seek the counsel of a licensed professional.
Ready to Start Your Business?




